Surfer 13 ternary plot11/5/2022  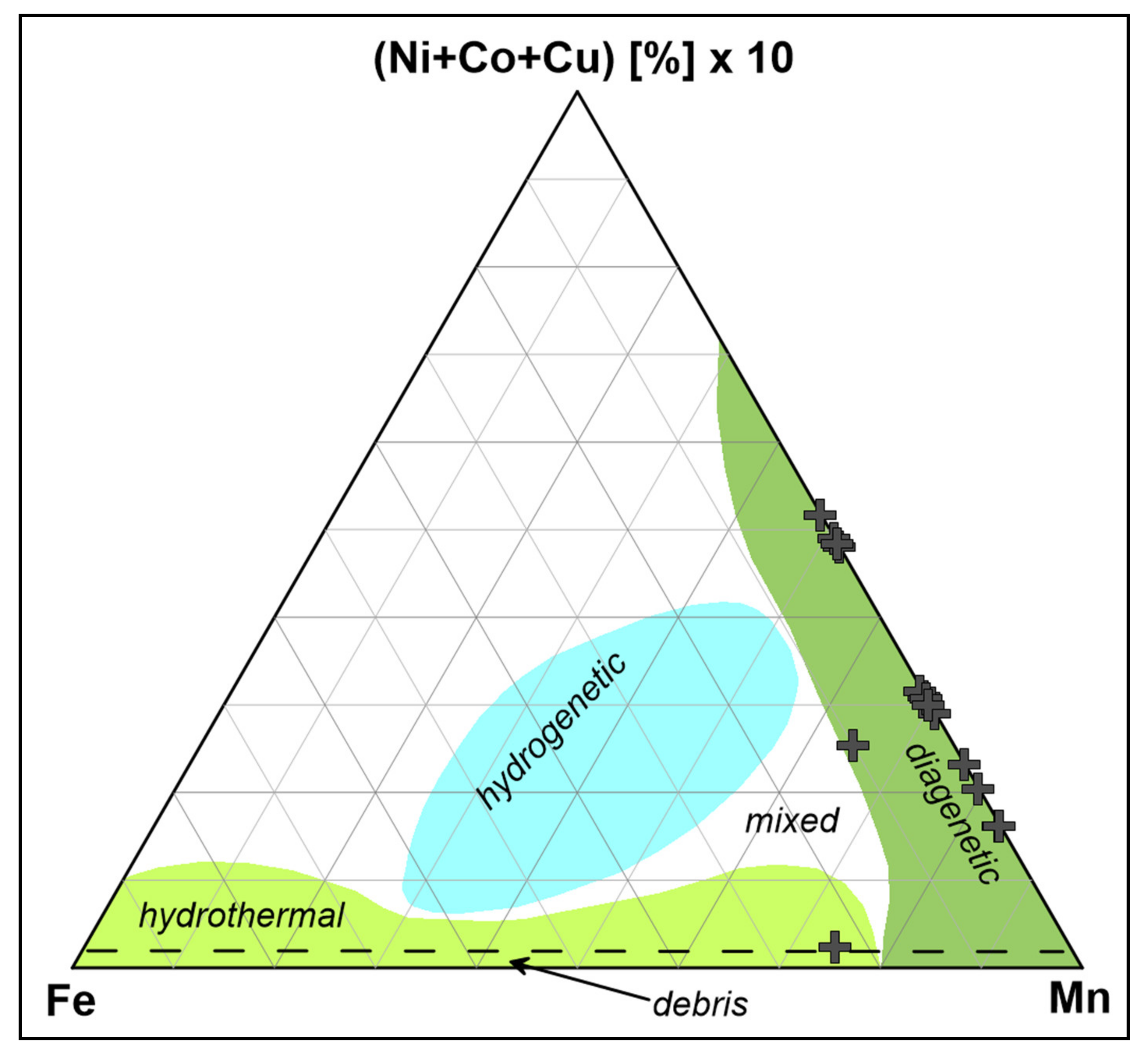
The axes crossover location is now fixed at the origin (0,0), so as we move or pan the plot, the crossover location changes its position in the chart area, without changing its coordinates. There are many ways to define the proportionality constant of Henry's law, which can be subdivided into two fundamental types: One possibility is to put the aqueous phase into the numerator and the gaseous phase into the denominator ("aq/gas").X and Y-axis location at origin: axes crossover fixed at (0,0) ( March 2016) ( Learn how and when to remove this template message) Unsourced material may be challenged and removed. Please help improve this article by adding citations to reliable sources. This section needs additional citations for verification. 
Fundamental types and variants of Henry's law constants To avoid this injury the diver must ascend slow enough that the excess dissolved gas is carried away by the blood and released into the lung gas. If the supersaturation is too great, bubbles may form and grow, and the presence of these bubbles can cause blockages in capillaries, or distortion in the more solid tissues which can cause damage known as decompression sickness. When ascending the diver is decompressed and the solubility of the gases dissolved in the tissues decreases accordingly. Solubility of gases increase at depth in accordance with Henry's law, so the body tissues take on more gas over time until saturated for the depth. In underwater diving Gas is breathed at the ambient pressure which increases with depth due to the hydrostatic pressure. For climbers or people living at high altitude Concentration of OĢ in the blood and tissues is so low that they feel weak and are unable to think properly, a condition called hypoxia. On opening the bottle to atmospheric pressure, solubility decreases and the gas bubbles are released from the liquid. Henry's law follows as a consequence if this ratio is a constant for each gas at a given temperature." Applications of Henry's law In production of carbonated beverages Under high pressure, solubility of COĢ increases.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
